Retroviral envelope proteins mediate receptor recognition and membrane fusion during early infection. Endogenous envelope proteins may have kept, lost or modified their original function during evolution. This endogenous envelope protein has retained its original fusogenic properties and participates in trophoblast fusion during placenta morphogenesis.
SU mediates receptor recognition. This interaction triggers the refolding of the transmembrane protein (TM) and is thought to activate its fusogenic potential by unmasking its fusion peptide (By similarity). Seems to recognize the type D mammalian retrovirus receptors SLC1A4 and SLC1A5, as it induces fusion of cells expressing these receptors in vitro.
The transmembrane protein (TM) acts as a class I viral fusion protein. Under the current model, the protein has at least 3 conformational states: pre-fusion native state, pre-hairpin intermediate state, and post-fusion hairpin state. During viral and target cell membrane fusion, the coiled coil regions (heptad repeats) assume a trimer-of-hairpins structure, positioning the fusion peptide in close proximity to the SLCterminal region of the ectodomain. The formation of this structure appears to drive apposition and subsequent fusion of membranes.
Function:
Retroviral envelope proteins mediate receptor recognition and membrane fusion during early infection. Endogenous envelope proteins may have kept, lost or modified their original function during evolution. This endogenous envelope protein has retained its original fusogenic properties and participates in trophoblast fusion during placenta morphogenesis.
SU mediates receptor recognition. This interaction triggers the refolding of the transmembrane protein (TM) and is thought to activate its fusogenic potential by unmasking its fusion peptide (By similarity). Seems to recognize the type D mammalian retrovirus receptors SLC1A4 and SLC1A5, as it induces fusion of cells expressing these receptors in vitro.
The transmembrane protein (TM) acts as a class I viral fusion protein. Under the current model, the protein has at least 3 conformational states: pre-fusion native state, pre-hairpin intermediate state, and post-fusion hairpin state. During viral and target cell membrane fusion, the coiled coil regions (heptad repeats) assume a trimer-of-hairpins structure, positioning the fusion peptide in close proximity to the SLCterminal region of the ectodomain. The formation of this structure appears to drive apposition and subsequent fusion of membranes (By similarity).
Subunit:
The mature envelope protein (Env) consists of a trimer of SU-TM heterodimers attached probably by a labile interchain disulfide bond. Interacts with the SLCtype lectin CD209/DSLCSIGN.
Subcellular Location:
Transmembrane protein: Cell membrane; Single-pass type I membrane protein (By similarity).
Surface protein: Cell membrane; Peripheral membrane protein (By similarity). Note=The surface protein is not anchored to the membrane, but localizes to the extracellular surface through its binding to TM (By similarity).
HERSLVW_7q21.2 provirus ancestral Env polyprotein: Virion (By similarity).
Tissue Specificity:
Expressed at higher level in placental syncytiotrophoblast. Expressed at intermediate level in testis. Seems also to be found at low level in adrenal tissue, bone marrow, breast, colon, kidney, ovary, prostate, skin, spleen, thymus, thyroid, brain and trachea. Both mRNA and protein levels are significantly increased in the brain of individuals with multiple sclerosis, particularly in astrocytes and microglia.
Post-translational modifications:
Specific enzymatic cleavages in vivo yield mature proteins. Envelope glycoproteins are synthesized as a inactive precursor that is heavily N-glycosylated and processed likely by furin in the Golgi to yield the mature SU and TM proteins. The cleavage site between SU and TM requires the minimal sequence [KR]-X-[KR]-R. The intracytoplasmic tail cleavage by the viral protease that is required for the fusiogenic activity of some retroviruses envelope proteins seems to have been lost during evolution.
The CXXC motif is highly conserved across a broad range of retroviral envelope proteins. It is thought to participate in the formation of a labile disulfide bond possibly with the CX6CC motif present in the transmembrane protein. Isomerization of the intersubunit disulfide bond to an SU intrachain disulfide bond is thought to occur upon receptor recognition in order to allow membrane fusion (By similarity).
Similarity:
Belongs to the gamma type-C retroviral envelope protein family. HERV class-I W env subfamily.
SWISS:
Q9UQF0
Gene ID:
30816
Database links:
Entrez Gene: 2086 Human
Entrez Gene: 30816 Human
Entrez Gene: 405754 Human
Omim: 604659 Human
SwissProt: O42043 Human
SwissProt: O71037 Human
SwissProt: P10267 Human
SwissProt: P60507 Human
SwissProt: P60508 Human
SwissProt: P61550 Human
SwissProt: P61565 Human
SwissProt: P61566 Human
SwissProt: P61567 Human
SwissProt: P61570 Human
SwissProt: Q14264 Human
SwissProt: Q69384 Human
SwissProt: Q902F8 Human
SwissProt: Q902F9 Human
SwissProt: Q96L62 Human
SwissProt: Q9N2J8 Human
SwissProt: Q9N2J9 Human
SwissProt: Q9N2K0 Human
SwissProt: Q9NX77 Human
SwissProt: Q9UKH3 Human
SwissProt: Q9UQF0 Human
Unigene: 250693 Human
Unigene: 631996 Human
合胞素(Syncytin)是一类由人俘获的逆转录病毒囊膜蛋白,与胎盘的形态发生中细胞滋养层到合胞滋养层的分化过程相关。Syncytin与人免疫缺陷病毒I型(HISLV1)囊膜蛋白(Env)在结构上具有相似的特点,二者可能具有相似的膜融合机制。
| Picture |
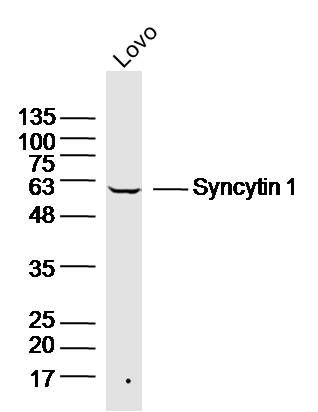
Sample:
Lovo(Human) Cell Lysate at 30 ug
Primary: Anti-Syncytin 1 (SL2962R) at 1/300 dilution
Secondary: IRDye800CW Goat Anti-Rabbit IgG at 1/20000 dilution
Predicted band size: 33/58 kD
Observed band size: 58kD
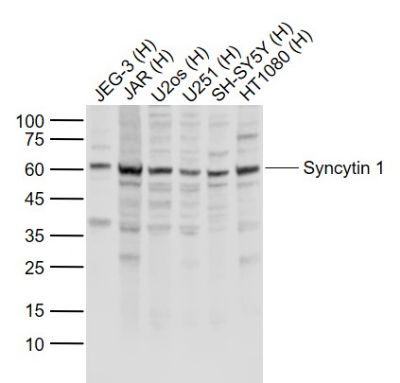
Sample:
Lane 1: JEG-3 (Human) Cell Lysate at 30 ug
Lane 2: JAR (Human) Cell Lysate at 30 ug
Lane 3: U2os (Human) Cell Lysate at 30 ug
Lane 4: U251 (Human) Cell Lysate at 30 ug
Lane 5: SH-SY5Y (Human) Cell Lysate at 30 ug
Lane 6: HT1080 (Human) Cell Lysate at 30 ug
Primary: Anti-Syncytin 1 (SL2962R) at 1/1000 dilution
Secondary: IRDye800CW Goat Anti-Rabbit IgG at 1/20000 dilution
Predicted band size: 58 kD
Observed band size: 60 kD
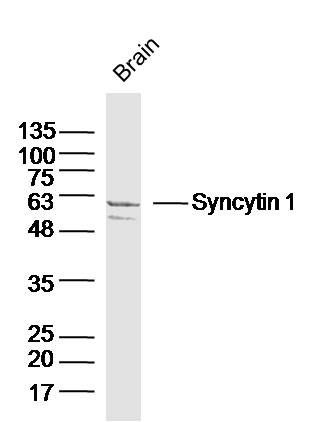
Sample:
Brain (Mouse) Lysate at 40 ug
Primary: Anti-Syncytin 1 (SL2962R) at 1/300 dilution
Secondary: IRDye800CW Goat Anti-Rabbit IgG at 1/20000 dilution
Predicted band size: 33/58 kD
Observed band size: 58kD
|
|
|