Vacuolar-type H+-ATPase (SLVATPase) is a multisubunit enzyme responsible for acidification of eukaryotic intracellular organelles. SLVATPases pump protons against an electrochemical gradient, while F-ATPases reverse the process, thereby synthesizing ATP. A peripheral V1 domain, which is responsible for ATP hydrolysis, and a integral V0 domain, which is responsible for proton translocation, compose SLVATPase. Nine subunits (A–H) make up the V1 domain and five subunits (a, d, c, c' and c") make up the V0 domain. Like F-ATPase, SLVATPase most likely operates through a rotary mechanism. SLVATPase D2 is a 350 amino acid protein that is expressed in kidney, lung and osteoclast. SLVATPase D2 has been implicated as a regulator of urine acidification, osteoclast fusion and bone formation. Furthermore, SLVATPase D2 has been identified as a dendritic cell marker.
Function:
Subunit of the integral membrane V0 complex of vacuolar ATPase. Vacuolar ATPase is responsible for acidifying a variety of intracellular compartments in eukaryotic cells, thus providing most of the energy required for transport processes in the vacuolar system. May play a role in coupling of proton transport and ATP hydrolysis (By similarity).
Subunit:
SLVATPase is a heteromultimeric enzyme composed of a peripheral catalytic V1 complex (components A to H) attached to an integral membrane V0 proton pore complex (components: a, c, c', c'' and d).
Subcellular Location:
Apical plasma membrane.
Tissue Specificity:
Kidney, osteoclast and lung.
Similarity:
Belongs to the SLVATPase V0D/AC39 subunit family.
SWISS:
Q8N8Y2
Gene ID:
245972
Database links:
Entrez Gene: 245972 Human
Entrez Gene: 242341 Mouse
Entrez Gene: 297932 Rat
SwissProt: Q8N8Y2 Human
SwissProt: Q80SY3 Mouse
SwissProt: Q5FVL0 Rat
Unigene: 43672 Human
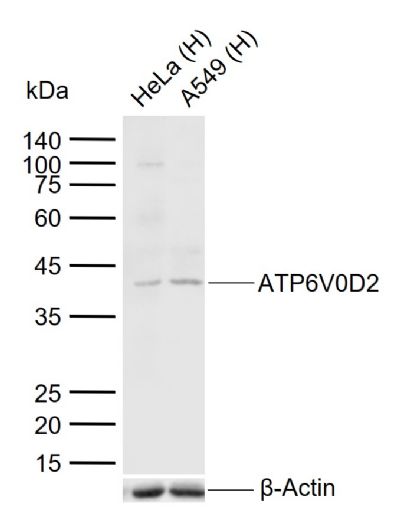
| Picture |
Sample:
Lane 1: Human HeLa cell lysates
Lane 2: Human A549 cell lysates
Primary: Anti-ATP6V0D2 (SL12548R) at 1/1000 dilution
Secondary: IRDye800CW Goat Anti-Rabbit IgG at 1/20000 dilution
Predicted band size: 40 kDa
Observed band size: 40 kDa
|
|
|